U.S. DOJ Reclassifies Marijuana Products to Schedule III
Here's what it means for you.
If you’re involved in the cannabis industry or a related sector, this reclassification could reshape your business landscape.
Why it matters
This shift in federal policy signals a potential easing of restrictions that could enhance medical research and market opportunities in the cannabis sector.
What happened (in 30 seconds)
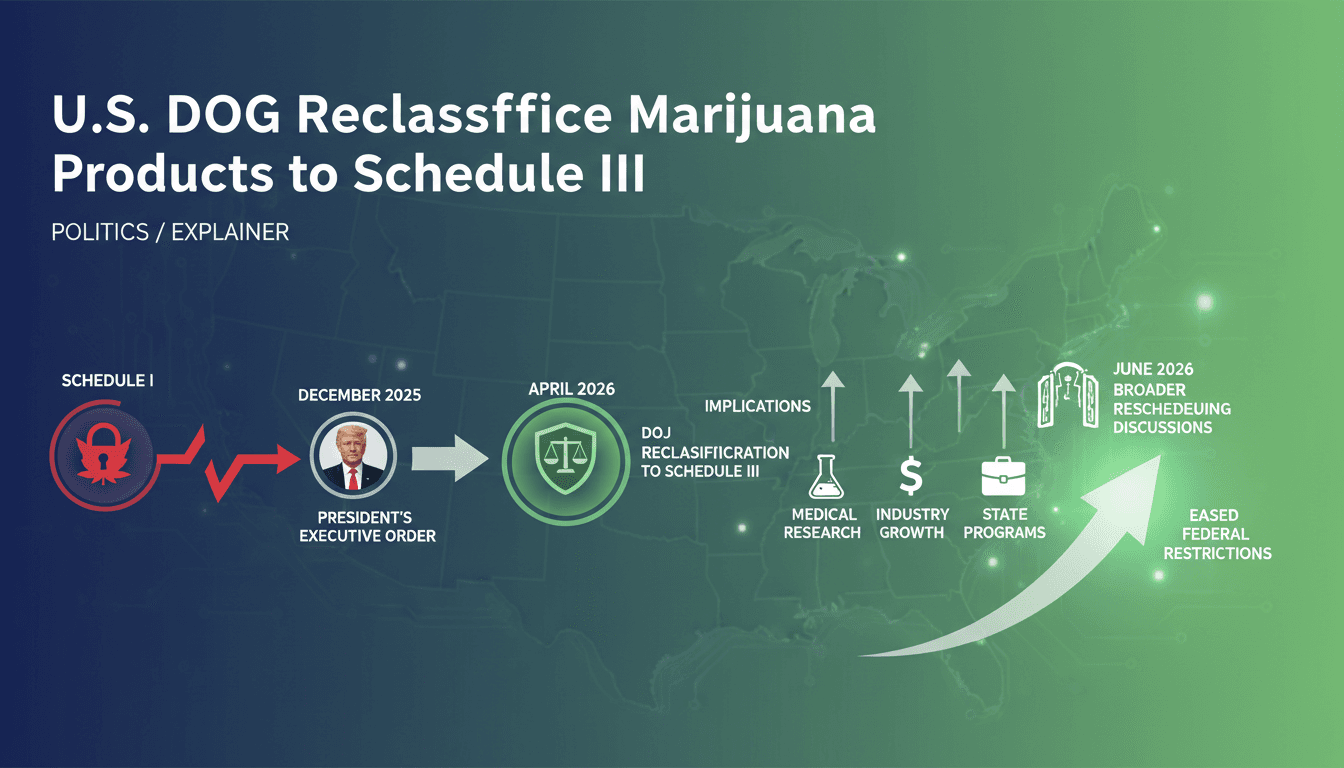
- On April 23, 2026, the U.S. Department of Justice reclassified FDA-approved and state-licensed medical marijuana products from Schedule I to Schedule III.
- Acting Attorney General Todd Blanche announced the immediate implementation, fulfilling President Trump's December 2025 executive order aimed at expanding medical research.
- An expedited hearing for broader marijuana rescheduling is set for June 29, 2026, while recreational marijuana remains federally illegal.
The context you actually need
- Marijuana has been classified as Schedule I since 1970, indicating high abuse potential and no accepted medical use, despite state-level legalization efforts.
- President Biden's administration proposed rescheduling in 2024, but the Trump administration halted those efforts upon taking office.
- The reclassification allows for tax relief under Section 280E for affected businesses, potentially easing financial burdens and improving banking access.
What's really happening
The reclassification of marijuana products by the U.S. Department of Justice (DOJ) marks a significant shift in federal drug policy, driven by a combination of political will and evolving public perception. Historically, marijuana has been classified as a Schedule I substance under the Controlled Substances Act, which denotes a high potential for abuse and no accepted medical use. This classification has long hindered research and development in the cannabis sector, despite growing evidence of its medical benefits and the legalization efforts at the state level.
The impetus for this change can be traced back to President Trump's executive order issued on December 18, 2025, which aimed to accelerate medical marijuana research and align federal policy with state regulations. This order recognized the need to adapt to the realities of state-level legalization and international treaty obligations. The DOJ's decision to reclassify FDA-approved and state-licensed medical marijuana products to Schedule III is a direct response to this executive directive, allowing for a more nuanced approach to cannabis regulation.
The immediate implications of this reclassification are significant. It eases federal restrictions on specific marijuana products, enabling greater access for patients and facilitating research into their medical applications. However, it does not legalize recreational marijuana at the federal level, maintaining a clear distinction between medical and recreational use. This nuanced approach reflects a growing recognition of the therapeutic potential of cannabis while still addressing concerns about abuse and public health.
Moreover, the reclassification opens the door for cannabis companies to benefit from tax relief under Section 280E, which has historically imposed heavy tax burdens on businesses operating in the cannabis space. This could lead to increased profitability and investment in the sector, as companies gain better access to banking services and financial resources. However, the initial market reaction was mixed, with cannabis stocks experiencing a surge of 6-13% before retreating as investors assessed the limited immediate scope of the reclassification.
As the expedited hearing for broader rescheduling approaches on June 29, 2026, the cannabis industry is poised for further developments. The outcome of this hearing could determine the future trajectory of cannabis regulation in the U.S., potentially paving the way for more comprehensive reforms.
Who feels it first (and how)
- Cannabis companies: They may see increased investment opportunities and reduced tax burdens.
- Patients: Access to FDA-approved medical marijuana products could improve, enhancing treatment options.
- Investors: Those in the cannabis sector may experience volatility as the market adjusts to the new regulations.
- Healthcare providers: They may gain more resources for research and treatment options involving medical marijuana.
What to watch next
- June 29, 2026, hearing outcomes: This will determine the future of broader marijuana rescheduling and could signal further regulatory changes.
- Market reactions: Watch for stock performance of cannabis companies as they adapt to the new regulations and potential tax relief.
- State-level responses: Monitor how individual states adjust their cannabis policies in light of federal changes, particularly regarding recreational use.
The DOJ has reclassified FDA-approved and state-licensed medical marijuana products to Schedule III.
The expedited hearing on June 29, 2026, will lead to further discussions on broader marijuana rescheduling.
The long-term impact on recreational marijuana legalization at the federal level remains uncertain.
Insights by A47 Intelligence
Macro commentary, policy analysis, growth/inflation themes, and global outlooks.
"Contextual macro coverage that complements day-to-day market headlines."
— A47 Editor
US to loosen marijuana rules in major shift for $47 billion industry
The US is set to loosen marijuana regulations, marking a significant shift in federal policy for the $47 billion industry. This change is expected to involve the reclassification of marijuana into a less restrictive federal category, a move anticipat...
Global news coverage with extensive reporting on Middle Eastern conflicts and geopolitics.
"Al Jazeera is a Qatar-based broadcaster known for wide regional coverage and alternative perspectives."
— A47 Editor
US reclassifies some marijuana products as less dangerous drug
The U.S. has reclassified certain marijuana products as less dangerous drugs, marking a significant shift in drug policy aimed at reducing penalties and promoting legalization efforts. This decision reflects a broader trend towards the acceptance of ...
Comprehensive coverage of Middle Eastern and global issues.
"Al Jazeera is a prominent voice from the Global South, especially the Middle East, with an emphasis on underreported stories."
— A47 Editor
US reclassifies some marijuana products as less dangerous drug
The U.S. has reclassified certain marijuana products as less dangerous drugs, marking a significant shift in drug policy aimed at reducing penalties and promoting legalization efforts. This decision reflects a broader trend towards the acceptance of ...
24/7 international news from a French perspective in multiple languages.
"France 24 is viewed as a globally focused outlet with balanced coverage and a European perspective."
— A47 Editor
Trump administration reclassifies medical marijuana as a 'less-dangerous drug'
The Trump administration has reclassified state-licensed medical marijuana as a less-dangerous drug, a decision announced on April 23, 2026. This reclassification aims to ease research barriers and provide tax relief, marking a significant shift in U...
UK and global health news, medicine, and public health research.
"BBC News is widely regarded as a reputable international news organization, known for its impartial tone and public service mandate."
— A47 Editor
Trump administration reclassifies cannabis as less dangerous
The Trump administration has reclassified medical marijuana from Schedule I to Schedule III, indicating a significant policy shift aimed at reducing the regulatory burden on cannabis research and access. This change aligns medical marijuana with less...
International coverage of politics, culture, and current affairs.
"BBC News is widely regarded as a reputable international news organization, known for its impartial tone and public service mandate."
— A47 Editor
Trump administration reclassifies cannabis as less dangerous
The Trump administration has reclassified cannabis, specifically medical marijuana, moving it into a lower category of drugs. This change aims to facilitate increased research and access to medical marijuana, reflecting a shift in the administration'...
Business, investment, entrepreneurship, leadership, and innovation.
"Forbes is known for its coverage of business leaders, market trends, and entrepreneurial ventures with a pro-business editorial stance."
— A47 Editor
Trump’s Justice Department Could Reclassify Marijuana As A Safer Drug Wednesday
President Trump’s Justice Department is poised to reclassify marijuana as a safer drug, following a directive issued in a December executive order. This potential rescheduling could take place as early as Wednesday, signaling a significant shift in f...
Global markets, investing, and macroeconomics from a premier financial newsroom.
"Bloomberg is respected for in-depth financial reporting and data-driven analysis."
— A47 Editor
DOJ Expected to Ease Marijuana Controls as Soon as Wednesday
The US Justice Department is anticipated to reclassify marijuana into a less restrictive federal category as soon as Wednesday, according to sources familiar with the situation. This potential change reflects a significant shift in federal policy reg...