FDA to Accelerate Drug Trials Using AI and Real-Time Data
Here's what it means for you.
This initiative could lead to faster access to new medications for patients and a more efficient drug development process for pharmaceutical companies.
What happened
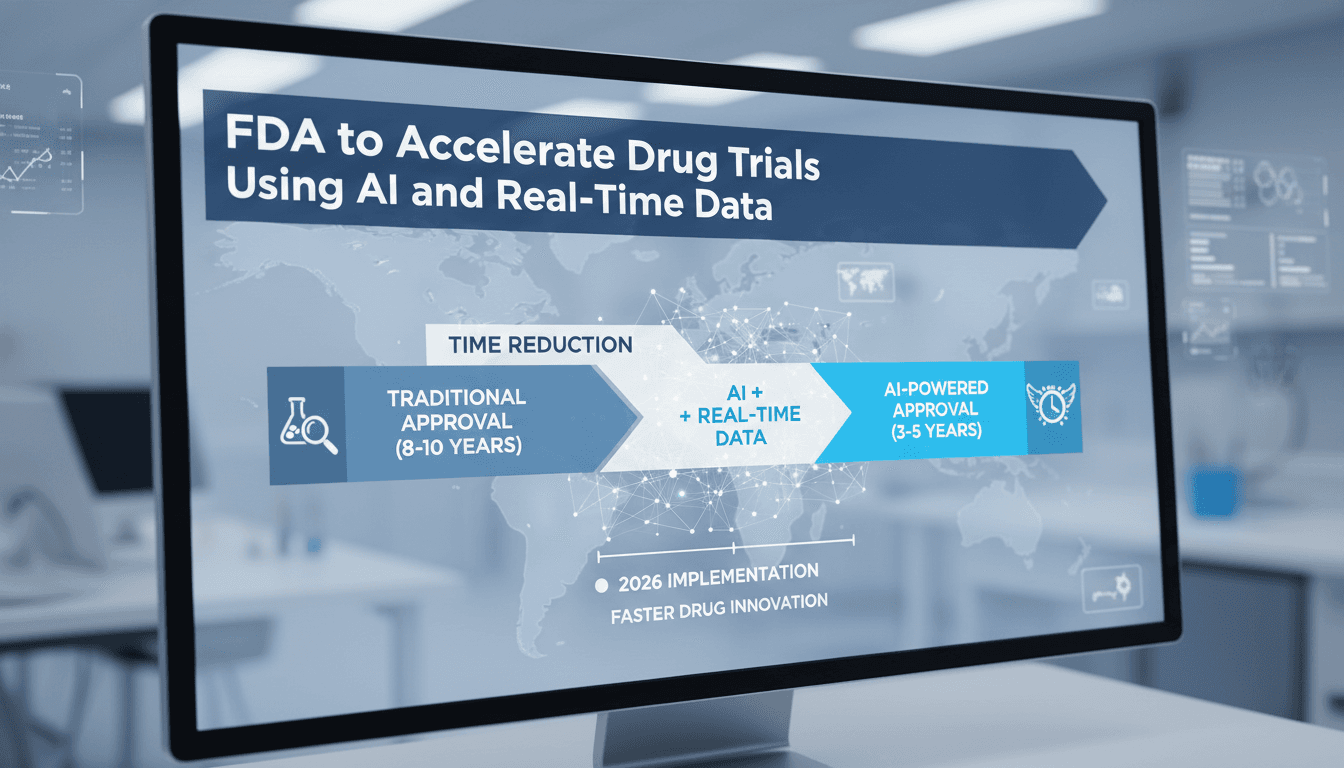
The FDA announced plans to accelerate drug trials using AI and real-time data.
The Context
- The FDA aims to improve visibility into ongoing clinical trials.
- AI will help streamline the data collection and submission process.
- This initiative is part of a broader effort to enhance drug development efficiency.
Takeaway
The FDA's use of AI could significantly shorten the timeline for bringing new medicines to market.
This article was generated by AI from 3 verified sources and reviewed by A47 editorial systems.
Technology business and AI-related headlines.
"Data-driven tech newsroom with global scope."
— A47 Editor
FDA Plans to Speed Up Drug Trials With Real-Time Data, AI
The US Food and Drug Administration (FDA) has announced plans to expedite the drug testing process by enhancing visibility into clinical trials through the use of real-time data and artificial intelligence. This initiative aims to reduce the time req...
Technology business news, market impacts, and innovation trends.
"Bloomberg is a premier financial and tech news provider, respected for its in-depth reporting and analytical rigor."
— A47 Editor
FDA Plans to Speed Up Drug Trials With Real-Time Data, AI
The US Food and Drug Administration (FDA) has announced plans to expedite the drug testing process by enhancing visibility into clinical trials through the use of real-time data and artificial intelligence. This initiative aims to reduce the time req...
Public health, medical research, healthcare policy, and pharmaceutical developments.
"Dow Jones is a major financial news provider with a professional tone and broad coverage, including specialized health-sector reporting."
— A47 Editor
FDA Turns to AI to Speed Up Clinical Trials
The U.S. Food and Drug Administration (FDA) is leveraging artificial intelligence to expedite the clinical trial process for new medications, aiming to streamline the extensive task of gathering and submitting research data. This initiative marks a s...
Tech business coverage, major deals, product launches, and Silicon Valley trends.
"WSJ’s tech section offers authoritative reporting on the intersection of technology and business, including exclusive industry analysis."
— A47 Editor
FDA Turns to AI to Speed Up Clinical Trials
The U.S. Food and Drug Administration (FDA) is implementing artificial intelligence (AI) to expedite the clinical trial process for new medicines. This initiative aims to streamline the often cumbersome task of collecting and submitting study data, p...